 
We can see the time is equal to seven seconds. Our graph right about there and we drop down and Molecules to one molecule so now our startingĬoncentration would be right here so what's half of that? That would be right here so we go over and we find this point on our graph. So the third half-life, we're going from two Twice as long as the first and each half-life is going to be twice as long as the one before it. So this makes sense,īoth looking at the graph and also thinking about theĮquation for our half-life. Half-life, which is one second, this is twice that, which ofĬourse would be two seconds. And we can see that this, that is twice this up here. This would be equal to two over k times the Alright so go back over here, this would be the initialĬoncentration of A divided by two. It's the initialĬoncentration divided by two. So the second, the second half-life would be equal to this would be one over the rate constant k but now our initial concentration, right, our initial concentration Half-life is one second here, let's go ahead and plug inįor the second half-life. Alright so if we say theįirst half-life is one second, so we say the first It's twice the first half-life and we can understand that by plugging in to our half-life equation over here. This time for our half-life? This time our half-life Graph would be right there and we drop down and we can see we're at time is equal to three. On our graph, right? So that point on our So here's half of this and if we go over, we can find this point The initial concentration divided by four now. 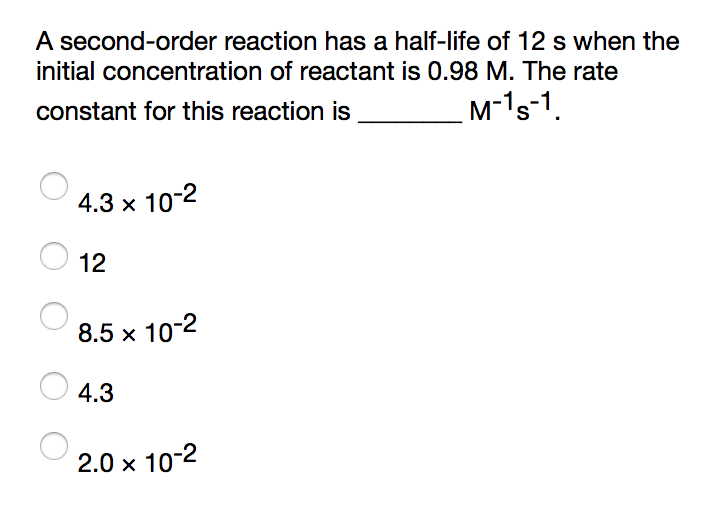
Starting concentration now, what's half of that? So on our graph half of How long does it take to do that? Well if this is our How long does it takeįor the second half-life? So how long does it take to go from four molecules to two molecules? Let me go ahead and changeĬolor and be consistent here. So we find that point on our graph and we drop down to here on the x-axis and it took one second. That would be the initialĬoncentration of A divided by two. The initial concentration, half that would be right here. To go from eight to four? Alright we can find that on our graph. Alright so we're left with four molecules so I'll draw those in there and how long did it take Alright, so if we waitįor the concentration to decrease to half its initial, what are we left with? We were left with four molecules. With eight particles that are eight molecules so here we have one, two, three, four, five, six, seven, eight. And let's just do a made-up reaction here. So here is our initial concentration of A. On the graph would be the initial concentration of A. So when time is equal to zero, alright, this point right here Of concentration versus time for a second order reaction so we can understand this conceptĪ little bit better. For a first order reaction we saw that the half-life was constant but here the half-life isn't constant because the half-life depends on the initial concentration of A. Notice this is very different for the half-life for a first order reaction. And so here's our equation for the half-life for a second order reaction. So we get the half-life is equal to one over k times the initial concentration of A. The initial concentration of A and that's equal to the rate constant k times the half-life. Initial concentration of A minus one over the initialĬoncentration of A? That would just be one over 
Initial concentration of A is equal to k t1/2. So one the left side this would just be two over the initial concentration of A minus one over the Initial concentration of A divided by two minus one over the initial concentration of A is equal to the rate constant k times the half-life. So it's half of our initial concentration. So our initialĬoncentration would be this. T 1/2 in for the time what's the concentration of A? Well using the definition for half-life, the concentration of A should be half of the initial concentration. Time is equal to t 1/2 so we're going to plug So if we're talking about the half-life so that would be when One over the concentration of A minus one over the initial concentration of A is equal to the rateĬonstant k times the time. Integrated rate equation for a second order reaction. We've also already talked about the integrated rate law or the Time that it takes for the concentration ofĪ reactant to decrease to half of its initial concentration. Remember half-life is symbolized by t 1/2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
